Osteoporosis- Biosimilar Technology and API
 PB Osteo-1010 is a biosimilar: Soon to be off patent. Osteoporosis product Biosimilar
PB Osteo-1010 is a biosimilar: Soon to be off patent. Osteoporosis product Biosimilar
Technology is product Biosimilar Technology.
PB-osteo-1010 is a recombinant form of the parathyroid hormone whose molecular weight of the final API is 4117 g/mol. PB-osteo-1010 has been developed for use in post-menopausal women with Osteoporosis possessing a high risk of fracture or with a history of Osteoporosis fracture. Increased bone mass in men with Osteoporosis and patients with multiple risk of fracture have also been taken into account during development.
The National Osteoporosis Foundation has estimated that 44 million people are currently experiencing the effect of Osteoporosis or Osteopenia. By 2020, this will have risen to more than 61 million people. Women are affected in greater numbers as they have a lower peak bone density.
Paras Biopharmaceuticals Finland Oy has successfully achieved a unique and noble process to obtain high expression.
| Product status | » SDS-PAGE High expressions. » Separation and purification of Diabrid achieved. » Confirmation through mass spectrometry. |
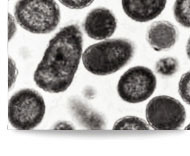
| Technology status | 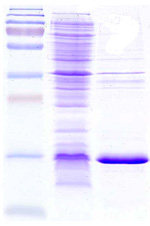 Soon ready to be out licensed for scale up production. Soon ready to be out licensed for scale up production.Recombinant stable E. Coli Clone exhibit deposits of the Osteoporosis product inside the cells (EM@39000-Paras Rights Reserved). SDS-PAGE-Gel image (left) showing marker (lane1), homogenization broth (lane2) and separation of product in Diabrids (lane3). |
Paras Biopharmaceuticals
Finland Oy,
Kiviharjunlenkki 10, OULU,
FI-90220 Finland
P: +358 442709462
Dr Ashesh Kumar
E: kumar.ashesh@parasbiopharma.com
P: +358 (0) 400207380
Dr Mark Jackson
E: mark.jackson@parasbiopharma.com
P:+358 (0) 442905993
Skype: Paras.Finland
This website uses cookies; by continuing to use this page, you consent to their use. I Understand About Cookies.